|
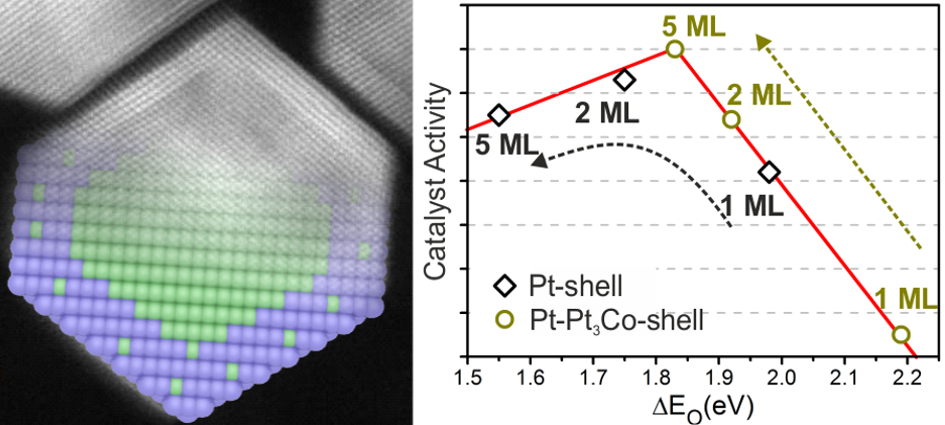
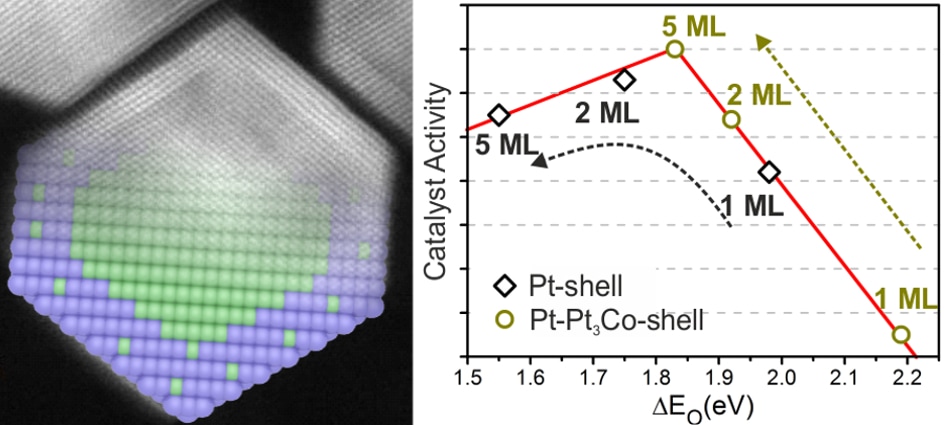
This time, we report the synthesis of truncated cuboctahedral Pt3Co-Pt core-shell nanoparticles. These nanoparticles are composed of a complete Pt monolayer skin, followed by layers with a ~78 At% of Pt, in a Pt3Co configuration, and finally a Co rich core with just 64 At% of Pt. This specific configuration exhibit an electrocatalytic activity ~6 times higher than commercial 30%-Pt/Vulcan. This work has been published in the Journal of Catalysis Science & Technology, and can be downloaded here. Guangzhi Hu, Eduardo Gracia-Espino, Robin Sandström, Tiva Sharifi, Shaodong Cheng, Hangjia Shen, Chuanyi Wang, Shaojun Guo, Guang Yang, Thomas Wågberg. Catal. Sci.Technol., 2015, DOI: 10.1039/C5CY01128K AbstractPtM-based core-shell nanoparticles are a new class of active and stable nanocatalysts to promote oxygen reduction reaction (ORR), however, the understanding at atomistic level of their high electrocatalytic performance for ORR is still a great challenge. Herein, we report a synthesis of highly ordered, and homogeneous truncated cuboctahedral Pt3Co-Pt core-shell nanoparticles (cs-Pt3Co). By combining atomic resolution electron microscopy, X-ray photoelectron spectroscopy, extensive first-principles calculations, and many other characterization techniques, we conclude that the cs-Pt3Co nanoparticles are composed of a complete or nearly complete Pt monolayer skin, followed by a secondary shell containing 5-6 layers with a ~78 At% of Pt, in a Pt3Co configuration, and finally a Co rich core with 64 At% of Pt. Only this particular structure is consistent with the very high electrocatalytic activity of cs-Pt3Co nanoparticles for ORR, which is about 6 times higher than commercial 30%-Pt/Vulcan, and 5 times more active than non-faceted (spherical) alloy Pt3Co nanoparticles. Our study gives an important insight into the atomistic design and understanding of advanced bimetallic nanoparticles for ORR catalysis and other important industrial catalytic applications.
0 Comments
Your comment will be posted after it is approved.
Leave a Reply. |
Nano for Energy group
Categories
All
Featured publications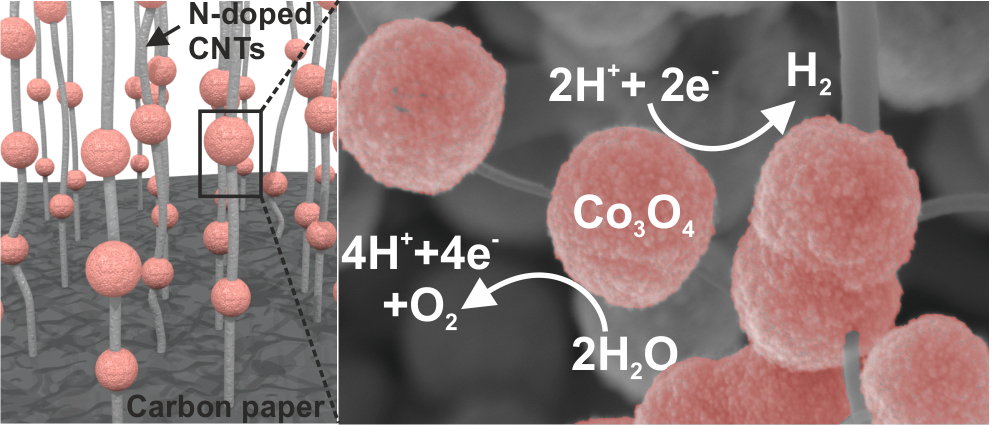
Comprehensive Study of an Earth-Abundant Bifunctional 3D Electrode for Efficient Water Electrolysis in Alkaline Medium.
ACS Appl. Mater. Interfaces, 2015, 7, 28148 
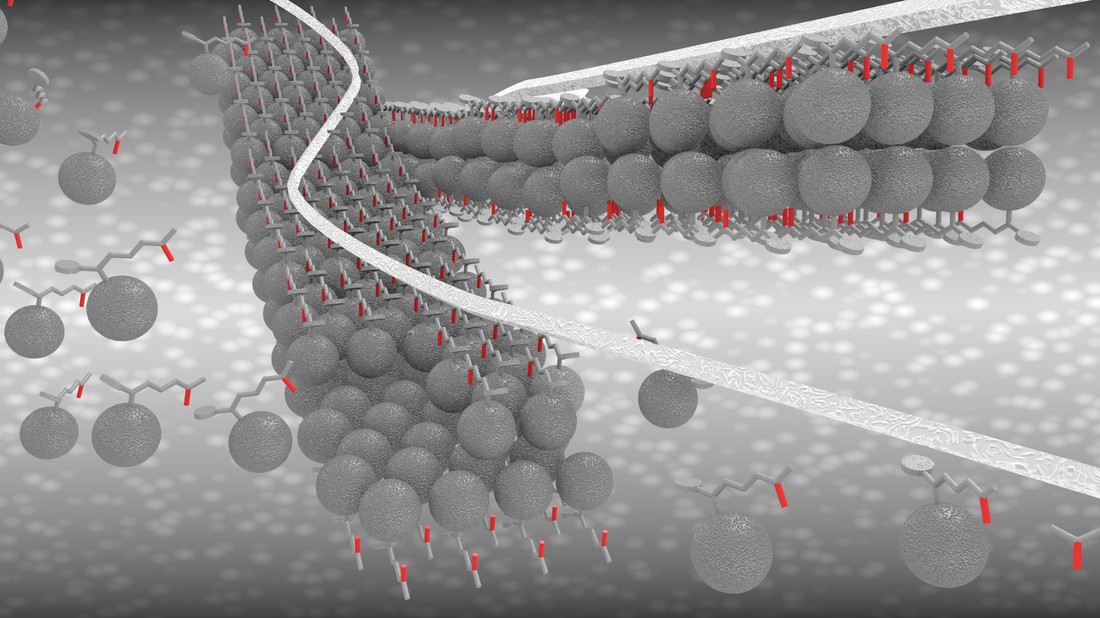
Fabrication of One-Dimensional Zigzag [6,6]-Phenyl-C61-Butyric Acid Methyl Ester Nanoribbons from Two-Dimensional Nanosheets.
ACS Nano, 2015, 9, 10516 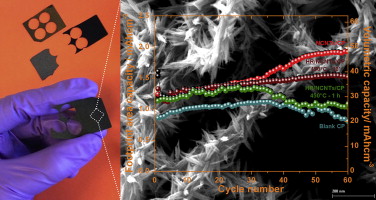
Hierarchical self-assembled structures based on nitrogen-doped carbon nanotubes as advanced negative electrodes for Li-ion batteries and 3D microbatteries.
J. P. Sources, 2015, 279, 581 .Self-Assembly Synthesis of Decorated Nitrogen-Doped Carbon Nanotubes with ZnO Nanoparticles: Anchoring Mechanism and the Effects of Sulfur.
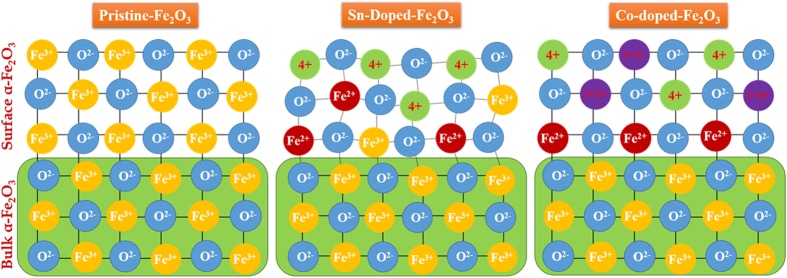
J. Phys. Chem. C, 120, 27849 (2016) Sn/Be Sequentially co-doped Hematite Photoanodes for Enhanced Photoelectrochemical Water Oxidation: Effect of Be2+ as co-dopant.
Sci Rep. 2016; 6: 23183. Atomistic understanding of the origin of high oxygen reduction electrocatalytic activity of cuboctahedral Pt3Co–Pt core–shell nanoparticles.
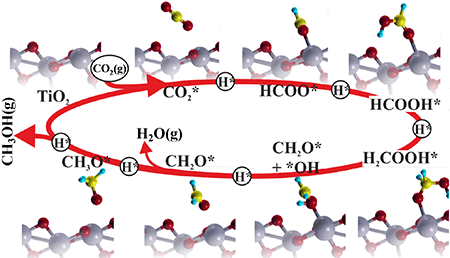
Catal. Sci. Technol., 2016, 6, 1393-1401 Photocatalytic reduction of CO2 with H2O over modified TiO2 nanofibers: Understanding the reduction pathway.
Nano Res. (2016) 9: 1956. |
 RSS Feed
RSS Feed