|
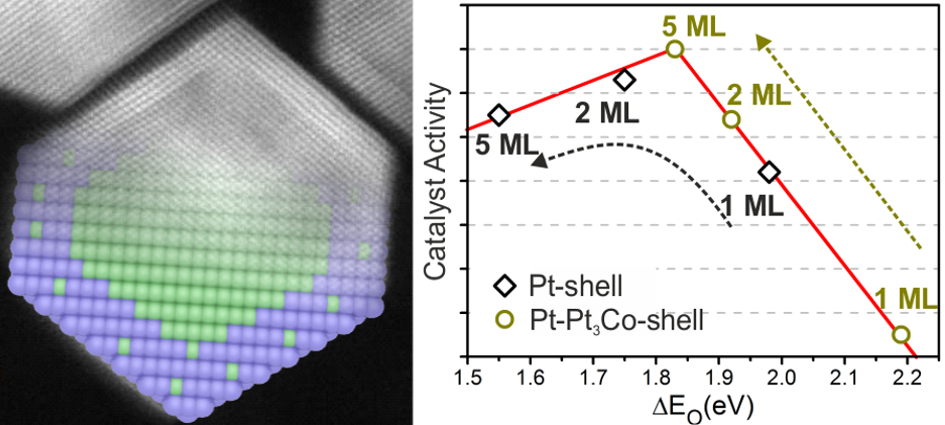
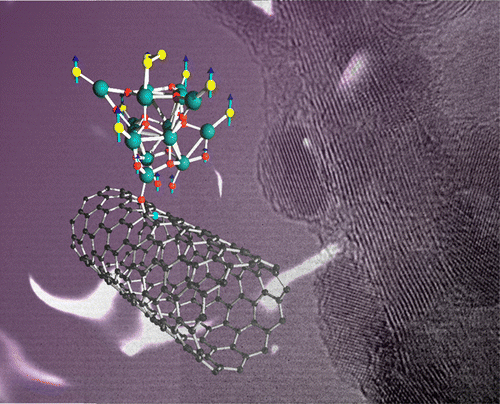
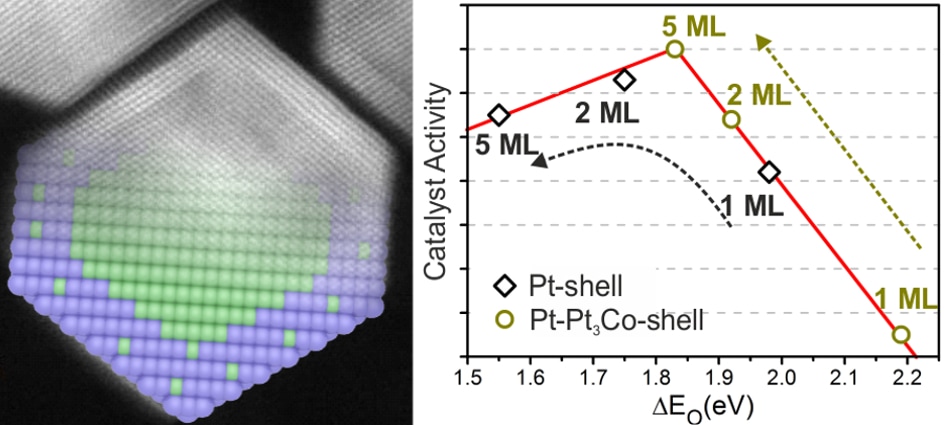
This time, we report the synthesis of truncated cuboctahedral Pt3Co-Pt core-shell nanoparticles. These nanoparticles are composed of a complete Pt monolayer skin, followed by layers with a ~78 At% of Pt, in a Pt3Co configuration, and finally a Co rich core with just 64 At% of Pt. This specific configuration exhibit an electrocatalytic activity ~6 times higher than commercial 30%-Pt/Vulcan. This work has been published in the Journal of Catalysis Science & Technology, and can be downloaded here. Guangzhi Hu, Eduardo Gracia-Espino, Robin Sandström, Tiva Sharifi, Shaodong Cheng, Hangjia Shen, Chuanyi Wang, Shaojun Guo, Guang Yang, Thomas Wågberg. Catal. Sci.Technol., 2015, DOI: 10.1039/C5CY01128K AbstractPtM-based core-shell nanoparticles are a new class of active and stable nanocatalysts to promote oxygen reduction reaction (ORR), however, the understanding at atomistic level of their high electrocatalytic performance for ORR is still a great challenge. Herein, we report a synthesis of highly ordered, and homogeneous truncated cuboctahedral Pt3Co-Pt core-shell nanoparticles (cs-Pt3Co). By combining atomic resolution electron microscopy, X-ray photoelectron spectroscopy, extensive first-principles calculations, and many other characterization techniques, we conclude that the cs-Pt3Co nanoparticles are composed of a complete or nearly complete Pt monolayer skin, followed by a secondary shell containing 5-6 layers with a ~78 At% of Pt, in a Pt3Co configuration, and finally a Co rich core with 64 At% of Pt. Only this particular structure is consistent with the very high electrocatalytic activity of cs-Pt3Co nanoparticles for ORR, which is about 6 times higher than commercial 30%-Pt/Vulcan, and 5 times more active than non-faceted (spherical) alloy Pt3Co nanoparticles. Our study gives an important insight into the atomistic design and understanding of advanced bimetallic nanoparticles for ORR catalysis and other important industrial catalytic applications.
0 Comments
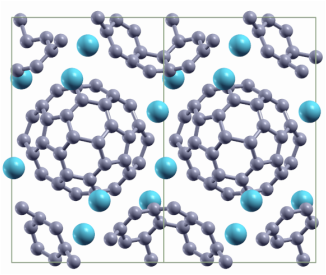
Here we performed calorimetric measurements of Na4C60 and Li4C60 crystals, the results are published in the Journal of Chemical Physics. Akira Inaba, Yuji Miyazaki, Paweł P. Michałowski, Eduardo Gracia-Espino, Bertil Sundqvist and Thomas Wågberg. J. Chem. Phys. 142, 164706 (2015) AbstractWe show specific heat data for Na4C60 and Li4C60 in the range 0.4-350 K for samples characterized by Raman spectroscopy and X-ray diffraction. At high temperatures, the two different polymer structures have very similar specific heats both in absolute values and in general trend. The specific heat data are compared with data for undoped polymeric and pristine C60. At high temperatures, a difference in specific heat between the intercalated and undoped C60 polymers of 100 J K−1 mol−1 is observed, in agreement with the Dulong-Petit law. At low temperatures, the specific heat data for Li4C60 and Na4C60 are modified by the stiffening of vibrational and librational molecular motion induced by the polymer bonds. The covalent twin bonds in Li4C60 affect these motions to a somewhat higher degree than the single intermolecular bonds in Na4C60. Below 1 K, the specific heats of both materials become linear in temperature, as expected from the effective dimensionality of the structure. The contribution to the total specific heat from the inserted metal ions can be well described by Einstein functions with T E = 386 K for Li4C60 and T E = 120 K for Na4C60, but for both materials we also observe a Schottky-type contribution corresponding to a first approximation to a two-level system with ΔE = 9.3 meV for Li4C60 and 3.1 meV for Na4C60, probably associated with jumps between closely spaced energy levels inside “octahedral-type” ionic sites. Static magnetic fields up to 9 T had very small effects on the specific heat below 10 K.
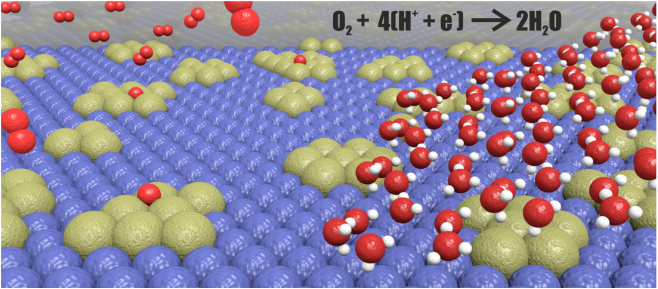
Our most recent article has been published in the journal of Nature Communications, and here, we report a unique novel Pd-W alloy with a Pd content of only 11 At%, which has a similar efficiency as a pure platinum catalyst. This excellent efficiency is explained by the unique morphology of the Pd-W nanoparticles. By advanced experimental and theoretical investigations, we show that the alloy is composed of metallic Pd-islands embedded in a W matrix. The size of the islands are about 1 nm in diameter and are composed of 10-20 atoms that are segregated to the surface. The unique formation of the material is based on the synthesis method, which is performed in an ordinary kitchen micro-wave oven. This study has been conducted in “The artificial leaf” project which is funded by Knut and Alice Wallenberg foundation to physicist, chemists, and plant science researchers at Umeå University. Guangzhi Hu, Florian Nitze, Eduardo Gracia-Espino, Jingyuan Ma, Hamid Reza Barzegar, Tiva Sharifi, Xueen Jia, Andrey Shchukarev, Lu Lu, Chuansheng Ma, Guang Yang, and Thomas Wågberg AbstractThe sluggish kinetics of the oxygen reduction reaction at the cathode side of proton exchange membrane fuel cells is one major technical challenge for realizing sustainable solutions for the transportation sector. Finding efficient yet cheap electrocatalysts to speed up this reaction therefore motivates researchers all over the world.
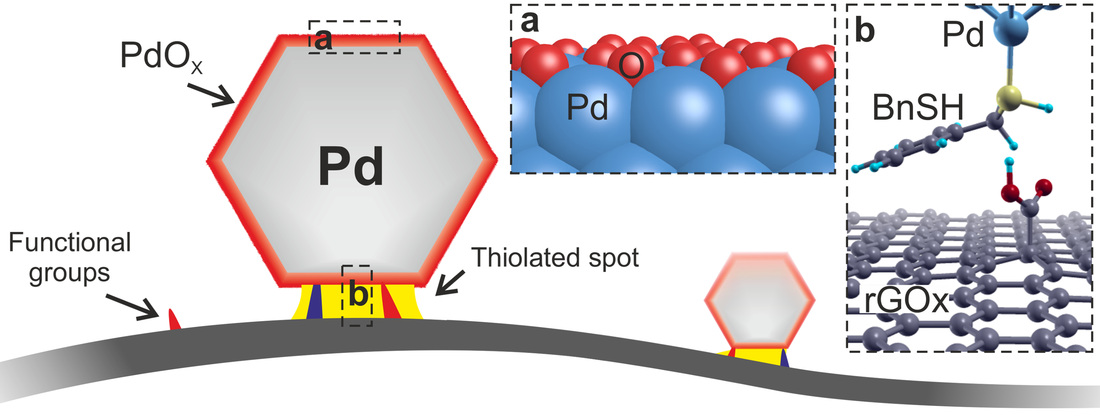
Here we demonstrate an efficient synthesis of palladium–tungsten bimetallic nanoparticles supported on ordered mesoporous carbon. Despite a very low percentage of noble metal (palladium:tungsten=1:8), the hybrid catalyst material exhibits a performance equal to commercial 60% platinum/Vulcan for the oxygen reduction process. The high catalytic efficiency is explained by the formation of small palladium islands embedded at the surface of the palladium–tungsten bimetallic nanoparticles, generating catalytic hotspots. The palladium islands are ~1 nm in diameter, and contain 10–20 palladium atoms that are segregated at the surface. Our results may provide insight into the formation, stabilization and performance of bimetallic nanoparticles for catalytic reactions. This is our most recent work published in the Journal of American Chemical Society. Here we studied the nanoscale interactions of reduced graphene oxide (rGOx) homogeneously decorated with small palladium nanoclusters (2.3 ± 0.3 nm). The Pd nanoparticles anchored to the rGOx-surface exhibit high crystallinity and are consistent with six-shell cuboctahedral and icosahedral clusters containing ∼600 Pd atoms. We also performed ab initio simulations to understand the electronic properties of the graphene−nanoparticle hybrid system. This article has been published as an open access, so here you can download the published version of the article. Eduardo Gracia-Espino, Guangzhi Hu, Andrey Shchukarev, and Thomas Wågberg. Abstract Studies on noble-metal-decorated carbon nanostructures are reported almost on a daily basis, but detailed studies on the nanoscale interactions for well-defined systems are very rare. Here we report a study of reduced graphene oxide (rGOx) homogeneously decorated with palladium (Pd) nanoclusters with well-defined shape and size (2.3 ± 0.3 nm). The rGOx was modified with benzyl mercaptan (BnSH) to improve the interaction with Pd clusters, and N,N-dimethylformamide was used as solvent and capping agent during the decoration process. The resulting Pd nanoparticles anchored to the rGOx-surface exhibit high crystallinity and are fully consistent with six-shell cuboctahedral and icosahedral clusters containing ∼600 Pd atoms, where 45% of these are located at the surface. According to X-ray photoelectron spectroscopy analysis, the Pd clusters exhibit an oxidized surface forming a PdOx shell. Given the well-defined experimental system, as verified by electron microscopy data and theoretical simulations, we performed ab initio simulations using 10 functionalized graphenes (with vacancies or pyridine, amine, hydroxyl, carboxyl, or epoxy groups) to understand the adsorption process of BnSH, their further role in the Pd cluster formation, and the electronic properties of the graphene−nanoparticle hybrid system. Both the experimental and theoretical results suggest that Pd clusters interact with functionalized graphene by a sulfur bridge while the remaining Pd surface is oxidized. Our study is of significant importance for all work related to anchoring of nanoparticles on nanocarbon-based supports, which are used in a variety of applications.
|
Nano for Energy group
Categories
All
Featured publications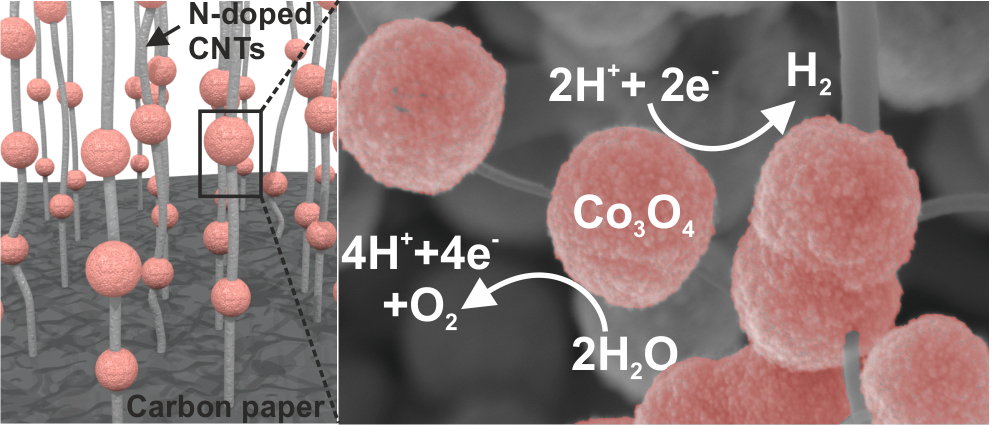
Comprehensive Study of an Earth-Abundant Bifunctional 3D Electrode for Efficient Water Electrolysis in Alkaline Medium.
ACS Appl. Mater. Interfaces, 2015, 7, 28148 
Fabrication of One-Dimensional Zigzag [6,6]-Phenyl-C61-Butyric Acid Methyl Ester Nanoribbons from Two-Dimensional Nanosheets.
ACS Nano, 2015, 9, 10516 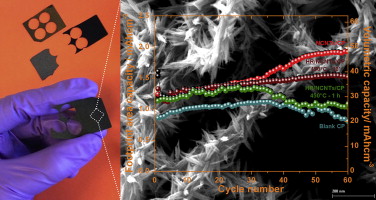
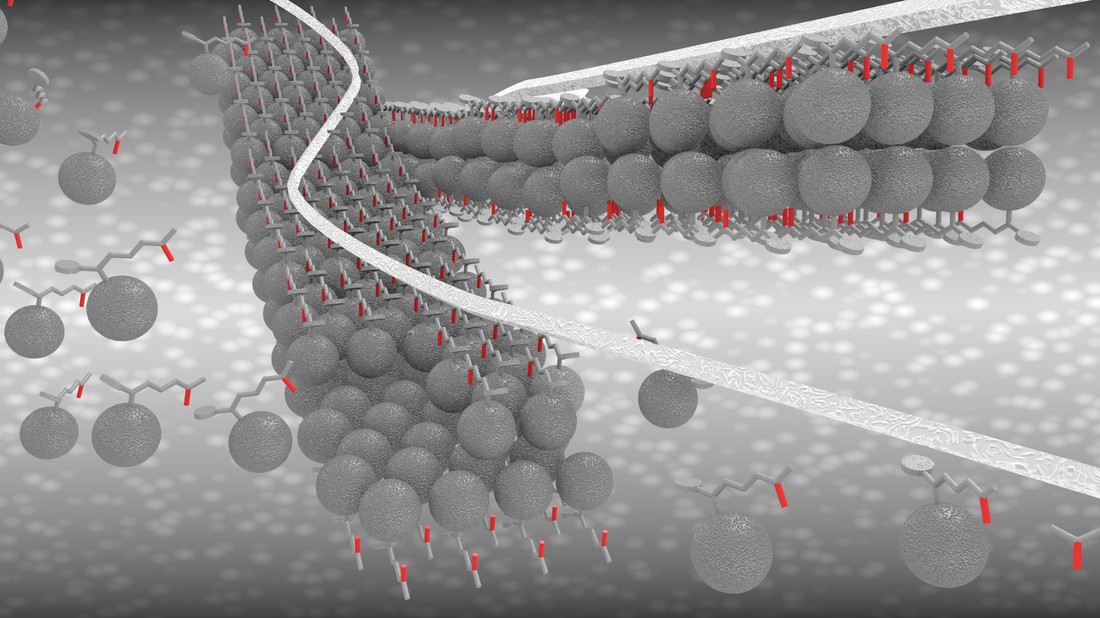
Hierarchical self-assembled structures based on nitrogen-doped carbon nanotubes as advanced negative electrodes for Li-ion batteries and 3D microbatteries.
J. P. Sources, 2015, 279, 581 .Self-Assembly Synthesis of Decorated Nitrogen-Doped Carbon Nanotubes with ZnO Nanoparticles: Anchoring Mechanism and the Effects of Sulfur.
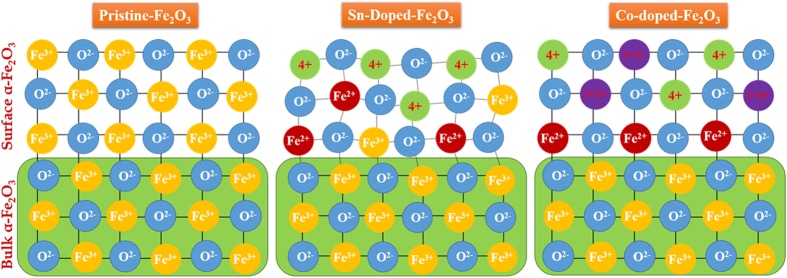
J. Phys. Chem. C, 120, 27849 (2016) Sn/Be Sequentially co-doped Hematite Photoanodes for Enhanced Photoelectrochemical Water Oxidation: Effect of Be2+ as co-dopant.
Sci Rep. 2016; 6: 23183. Atomistic understanding of the origin of high oxygen reduction electrocatalytic activity of cuboctahedral Pt3Co–Pt core–shell nanoparticles.
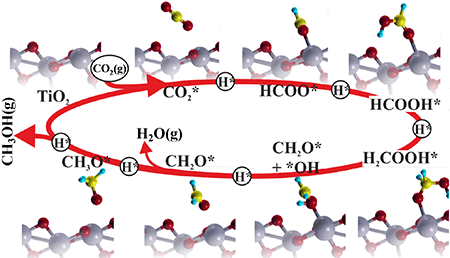
Catal. Sci. Technol., 2016, 6, 1393-1401 Photocatalytic reduction of CO2 with H2O over modified TiO2 nanofibers: Understanding the reduction pathway.
Nano Res. (2016) 9: 1956. |
 RSS Feed
RSS Feed