|
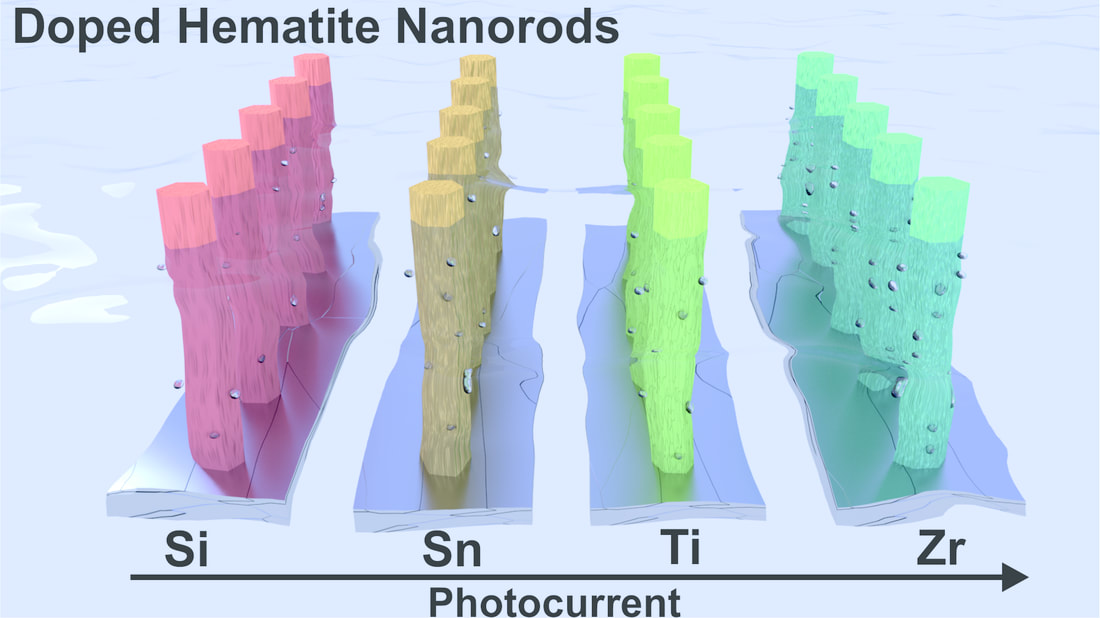
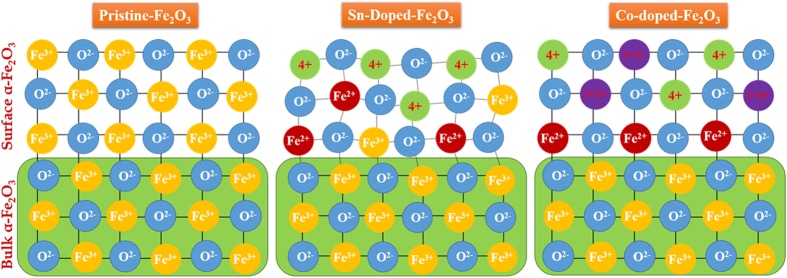
This time the influence of tetravalent dopants such as Si4+, Sn4+, Ti4+, and Zr4+ on hematite nanostructure for enhanced photoelectrochemical water splitting is reported. The photoactivity of the doped photoanodes at 1.23 V RHE follows the order Zr > Sn > Ti > Si. The work was performed in collaboration with Prof. Jum Suk Jang (Chonbuk National University, Korea), and the results are published in the journal of Applied Surface Science. A. Subramanian, E. Gracia-Espino, A. Annamalai, H. H. Lee, S Y. Lee, S. H. Choi, and J. S. Jang. Applied Surface Science (2017). DOI: 10.1016/j.apsusc.2017.09.042 Abstract In this paper, the influence of tetravalent dopants such as Si4+, Sn4+, Ti4+, and Zr4+ on the hematite (α-Fe2O3) nanostructure for enhanced photoelectrochemical (PEC) water splitting are reported. The tetravalent doping was performed on hydrothermally grown akaganeite (β-FeOOH) nanorods on FTO (fluorine-doped tin-oxide) substrates via a simple dipping method for which the respective metal-precursor solution was used, followed by a high-temperature (800° C) sintering in a box furnace. The photocurrent density for the pristine (hematite) photoanode is ∼0.81 mA/cm2 at 1.23 VRHE, with an onset potential of 0.72 VRHE; however, the tetravalent dopants on the hematite nanostructures alter the properties of the pristine photoanode. The Si4+-doped hematite photoanode showed a slight photocurrent increment without a changing of the onset potential of the pristine photoanode. The Sn4+- and Ti4+-doped hematite photoanodes, however, showed an anodic shift of the onset potential with the photocurrent increment at a higher applied potential. Interestingly, the Zr4+-doped hematite photoanode exhibited an onset potential that is similar to those of the pristine and Si4+-doped hematite, but a larger photocurrent density that is similar to those of the Sn4+- and Ti4+-doped photoanodes was recorded. The photoactivity of the doped photoanodes at 1.23 VRHE follows the order Zr > Sn > Ti > Si. The onset-potential shifts of the doped photoanodes were investigated using the Ab initio calculations that are well correlated with the experimental data. X-ray diffraction (XRD) and scanning-electron microscopy (FESEM) revealed that both the crystalline phase of the hematite and the nanorod morphology were preserved after the doping procedure. X-ray photoelectron spectroscopy (XPS) confirmed the presence of the tetravalent dopants on the hematite nanostructure. The charge-transfer resistance at the various interfaces of the doped photoanodes was studied using impedance spectroscopy. The doping on the hematite photoanodes was confirmed using the Mott-Schottky (MS) analysis.
0 Comments
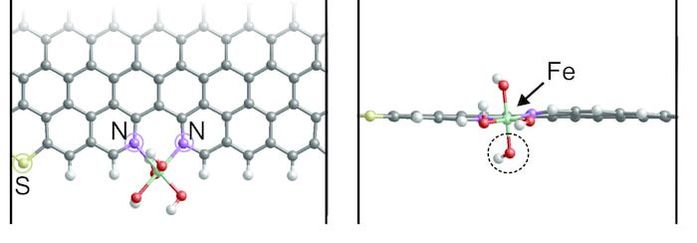
In this occasion, we investigated a sulfur-doped Fe-N-C (Fe/SNC) catalyst with a thiophene-like structure (C-S-C) that reduces the electron localization around the Fe center and improves the interaction with oxygenated species. The observed synergistic effect makes the Fe/SNC catalyst exhibits better ORR activity than sulfur free catalyst (Fe/NC) in 0.5 M H2SO4. The results were published in the journal of Angewandte Chemie International Edition. Hangjia Shen, Eduardo Gracia- Espino, Jingyuan Ma, Ketao Zang, Jun Luo, Le Wang, Sanshuang Gao, Xamxikamar Mamat, Guangzhi Hu, Thomas Wagberg, and Shaojun Guo. Angew. Chem. Int. Ed. (2017), DOI: 10.1002/anie.201706602 AbstractVarious advanced catalysts of sulfur doped Fe-N-C materials have been recently designed for oxygen reduction reaction (ORR), however, the enhanced activity is still controversial and usually attributed to differences in surface area, improved conductivity, or to uncertain synergistic effects. Here, a sulfur-doped Fe-N-C catalyst (denoted as Fe/SNC) derived via a template sacrificing method is presented. The incorporated S gives a thiophene-like structure (C-S-C), reduces the electron localization around the Fe center, improves the interaction with oxygenated species, and therefore facilitates the complete 4e- ORR in acid solution. This synergistic effect makes the Fe/SNC catalyst exhibits much better ORR activity than sulfur free catalyst (Fe/NC) in 0.5 M H2SO4.
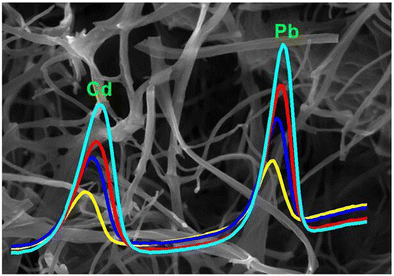
Carbon nanofibers were prepared by one-step carbonization of bacterial cellulose and used to detect Cd(II) and Pb(II). The detection limits are 0.38 μg·L−1 for Cd(II) and 0.33 μg·L−1 for Pb(II), respectively. The work was developed as a collaboration with Prof. Hu and Prod. Mamat (China). The results are published in the journal of Microchimica Acta. Danfeng Qin, Shanshuang Gao, Le Wang, Hangjia Shen, Nuerbiya Yalikun, Parviz Sukhrobov, Thomas Wågberg, Yujie Zhao, Xamxikamar Mamat, Guangzhi Hu. Microchimica Acta, 184, 2759–2766 (2017) Abstract The authors describe the preparation of carbon nanofibers (CNFs) with a three-dimensional network structure by one-step carbonization of bacterial cellulose at 800 °C. The 3D CNFs wrapped with Nafion polymer were cast on a glassy carbon electrode (GCE) which then enables sensitive detection of Cd(II) and Pb(II). Under optimized conditions and at typical stripping peaks of around −0.80 and −0.55 V (vs Ag/AgCl), the electrode exhibits high sensitivity and a wide analytical range of 2–100 μg·L−1 for both Cd(II) and Pb(II). The detection limits are 0.38 μg·L−1 for Cd(II) and 0.33 μg·L−1 for Pb(II), respectively. The modified GCE was successfully employed to the determination of trace amounts of Cd(II) and Pb(II) in both tap water and waste water.
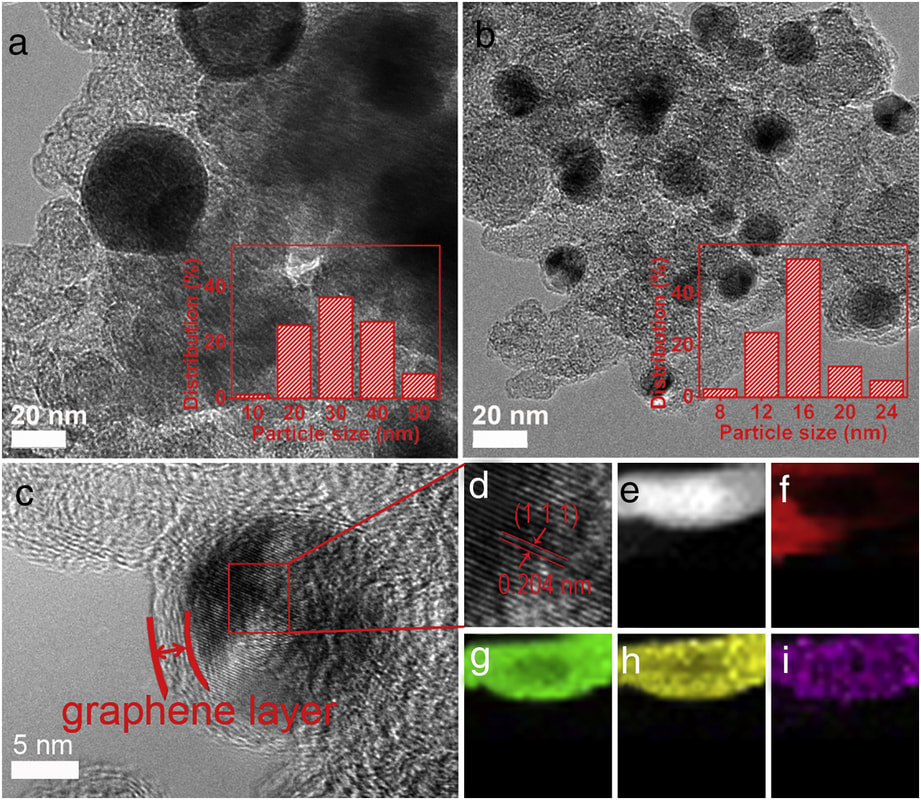
This work was performed in collaboration with Prof. Guangzhi Hu from the University of Chinese Academy of Sciences. Here we report the microwave-assisted synthesis of Mo-doped FeNi3 nanoparticles as excellent oxygen evolution electrocatalyst. Our results were published in the journal of Electrochemistry Communications. Hangjia Shen, Eduardo Gracia-Espino, Le Wang, Dan feng Qin, Sanshuang Gao, Xamxikamar Mamat, Wei Ren, Thomas Wågberg, Guangzhi Hu. Electrochem. Commun. 81, 116-119 (2017) Abstract Oxygen evolution reaction (OER) plays a pivotal role in water-splitting. Here, we report a facile method to synthesize multimetal supported on commercial carbon black via a time-saving microwave process. Crystalline FeNi3 nanoparticles homogeneously doped with Mo are formed via a microwave treatment and activated to metal oxyhydroxide in-situ during cyclic voltammetry test with overpotential of only 280 mV at 10 mA cm− 2 for OER in alkaline electrolyte, outperforming RuO2. Our synthesis methodology is a promising alternative for large-scale production, delivering a valuable contribution to catalyst preparation and electrocatalytic water oxidation research.
|
Nano for Energy group
Categories
All
Featured publications
Comprehensive Study of an Earth-Abundant Bifunctional 3D Electrode for Efficient Water Electrolysis in Alkaline Medium.
ACS Appl. Mater. Interfaces, 2015, 7, 28148 
Fabrication of One-Dimensional Zigzag [6,6]-Phenyl-C61-Butyric Acid Methyl Ester Nanoribbons from Two-Dimensional Nanosheets.
ACS Nano, 2015, 9, 10516 
Hierarchical self-assembled structures based on nitrogen-doped carbon nanotubes as advanced negative electrodes for Li-ion batteries and 3D microbatteries.
J. P. Sources, 2015, 279, 581 .Self-Assembly Synthesis of Decorated Nitrogen-Doped Carbon Nanotubes with ZnO Nanoparticles: Anchoring Mechanism and the Effects of Sulfur.
J. Phys. Chem. C, 120, 27849 (2016) Sn/Be Sequentially co-doped Hematite Photoanodes for Enhanced Photoelectrochemical Water Oxidation: Effect of Be2+ as co-dopant.
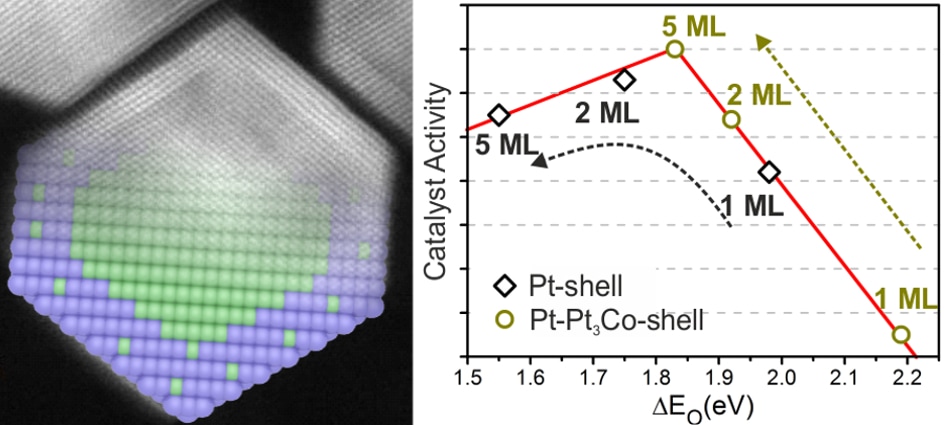
Sci Rep. 2016; 6: 23183. Atomistic understanding of the origin of high oxygen reduction electrocatalytic activity of cuboctahedral Pt3Co–Pt core–shell nanoparticles.
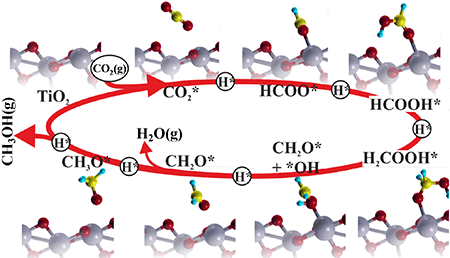
Catal. Sci. Technol., 2016, 6, 1393-1401 Photocatalytic reduction of CO2 with H2O over modified TiO2 nanofibers: Understanding the reduction pathway.
Nano Res. (2016) 9: 1956. |
 RSS Feed
RSS Feed