|
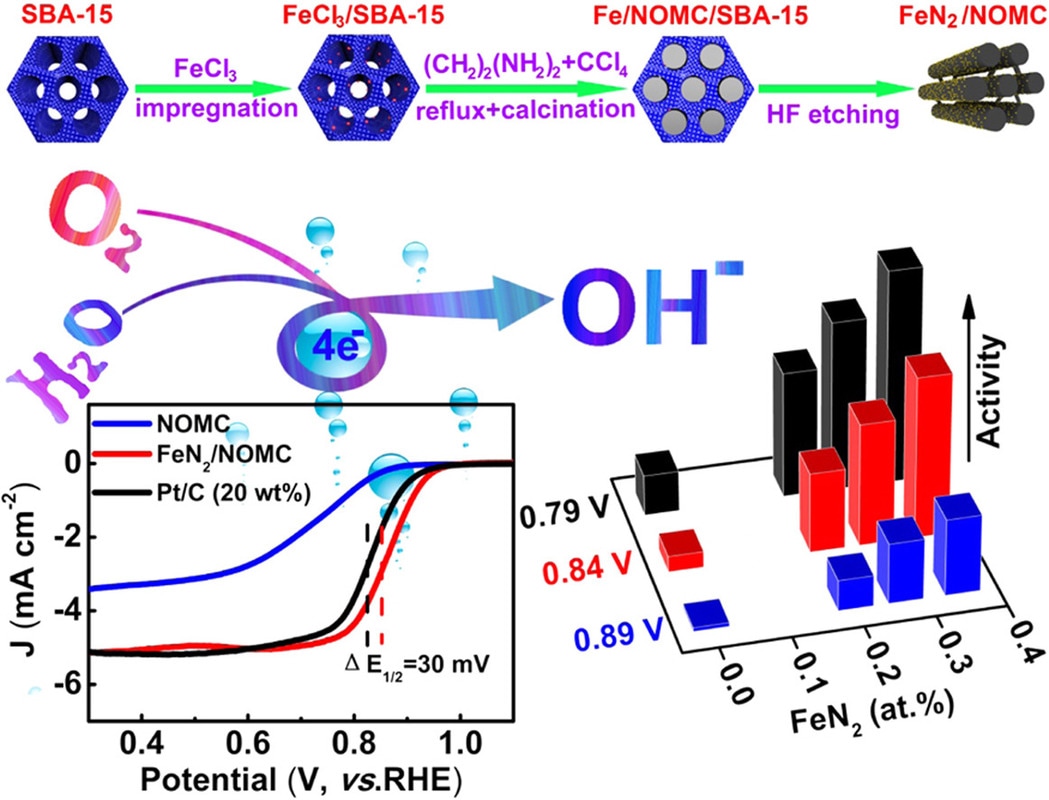
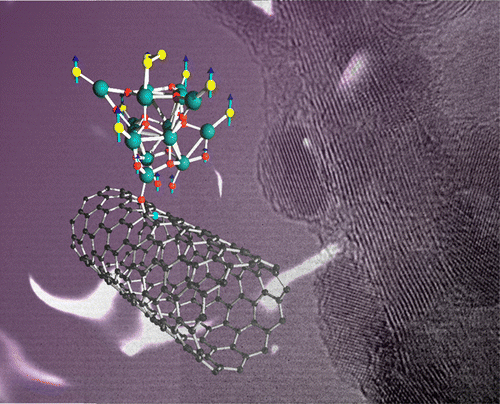
We now report a template casting strategy to easily introduce atomically dispersed FeN2 moieties onto the surface of N-doped ordered mesoporous carbon with extraordinary catalytic activity towards ORR. This work was carried out in collaboration with Prof. Guo (College of Engineering, Peking University) and Prof. Hu (Xinjiang Technical Institute of Physics and Chemistry). The results are published in the journal of Nano Energy. H. Shena, E. Gracia-Espino, J. Mac, H. Tang, X. Mamat, T. Wagberg, G. Hua, S. Guoe. Nano Energy, (2017) DOI: 10.1016/j.nanoen.2017.03.027 Abstract Earth-abundant materials with Fe-N-C centers have been identified as promising catalysts for oxygen reduction reaction (ORR), but these alternatives for Pt catalysts are usually the porphyrin-like FeN4 configuration. The density functional theory (DFT) calculations reveal that FeN2 outperforms FeN4 due to its lower interaction with ⁎O2 and ⁎OH intermediates and enhanced electron transport, however, achieving an optimum design of these earth-abundant materials with the enriched FeN2 catalytic centers is still a great challenge. Here, we report an intriguing template casting strategy to introduce a mass of atomically dispersed FeN2 moieties onto the surface of N-doped ordered mesoporous carbon for boosting ORR electrocatalysis. One of unique parts herein is to pre anchor Fe precursor on the surface of template (SBA-15) during catalyst synthesis, preventing Fe from penetrating into the carbon skeleton and facilitating the removal of excessive Fe-based particles during silica elimination by HF etching, resulting in a desirable model structure comprising only highly active atomically dispersed FeN2 sites, as confirmed by high-angle annular dark field-scanning transmission electron microscopy (HAADF-STEM), extended X-ray absorption fine structure (EXAFS) and Mößbauer spectroscopy analysis. The well-defined structure prompts us to understand the nature of the catalytic active sites, and to demonstrate that the catalyst activity is linearly proportional to the concentration of FeN2 sites. The obtained atomic electrocatalyst exhibits superior electrocatalytic performance for ORR with a more positive half-wave potential than that of Pt/C catalyst. We further establish a kinetic model to predict the ORR activity of these single-atom dispersed catalysts. The present work elaborates on a profound understanding for designing low-cost, highly efficient FeN2-based electrocatalyst for boosting ORR.
0 Comments
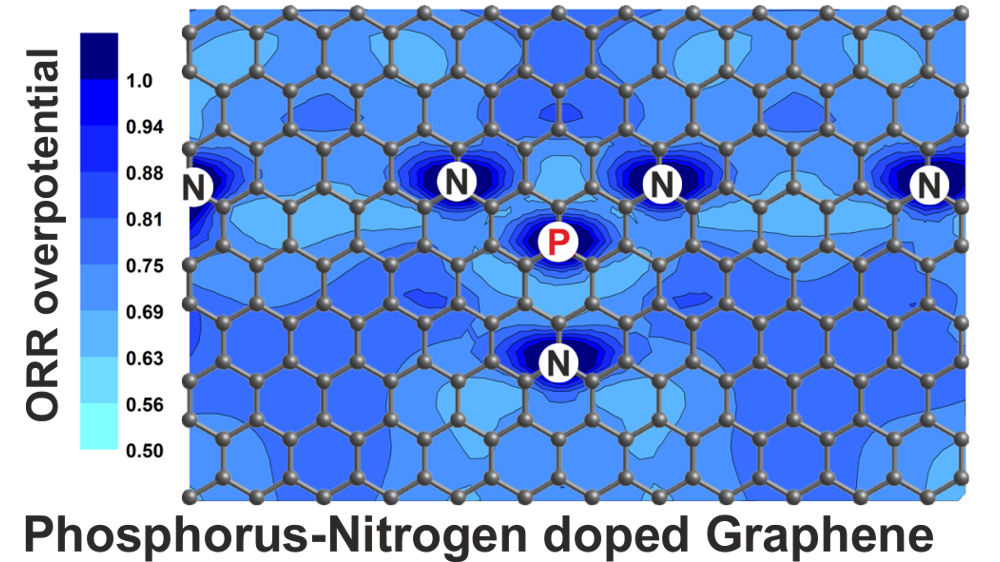
In our most recent publication we performed ab initio calculations to construct ORR overpotential maps and describe the availability and spatial distribution of catalytic active sites on phosphorus-nitrogen co-doped graphene. The results are published in the Journal of Physical Chemistry C. Eduardo Gracia-Espino. J. Phys. Chem. C, 120, 27849–27857 (2016) DOI: 10.1021/acs.jpcc.6b09425 Abstract Ab initio calculations are performed to investigate how the simultaneous introduction of phosphorus and nitrogen into graphene modifies the availability and spatial distribution of catalytic active sites for oxygen reduction reaction (ORR). A phosphoryl group (R3-P=O) is selected as a representative for the phosphorus doping, and the ORR is studied under alkaline conditions where a 4e- mechanism is used to determine the limiting step and overpotential (ηORR) along the entire graphene surface. A scanning procedure is used to construct ηORR maps for pristine-, N-, P-, and diverse PN co-doped graphenes. The results indicate that a single N (P) atom activates up to 17 (3) C atoms, while the simultaneous introduction of P and N activates up to 55 C atoms equivalent to 57% of the surface. Additionally, PN co-doped graphenes reveals that the relative location of both dopants has significant effects on the ORR performance, where a P-N separation distance of at least 4 Å minimize the localization of electronic states on the neighboring C atoms and improves the quantity and distribution of active sites. The results shows the importance of designing synthesis procedures to control the dopant concentration and spatial distribution to maximize the number of active sites. Furthermore, the ηORR maps reveal features that could be obtained by scanning tunneling microscopy allowing to experimentally identify and possibly quantify the catalytic active sites on carbon-based materials.
This time we report an artificial-leaf device where NiCo2O4 nanorods are used as bifunctional electrode that it can operate as both anode and cathode in the same alkaline solution. By driving two such identical electrodes with a perovskite photovoltaic assembly, a wired artificial-leaf device is obtained that features a Faradaic H2 evolution efficiency of 100%, and a solar-to-hydrogen conversion efficiency of 6.2%. This work was carried out in close collaboration with the research groups of Prof. Edman and Prof. Messinger. The article is published in the journal of Advanced Energy Materials as open access.
AbstractMolecular hydrogen can be generated renewably by water splitting with an “artificial-leaf device”, which essentially comprises two electrocatalyst electrodes immersed in water and powered by photovoltaics. Ideally, this device should operate efficiently and be fabricated with cost-efficient means using earth-abundant materials. Here, a lightweight electrocatalyst electrode, comprising large surface-area NiCo2O4 nanorods that are firmly anchored onto a carbon–paper current collector via a dense network of nitrogen-doped carbon nanotubes is presented. This electrocatalyst electrode is bifunctional in that it can efficiently operate as both anode and cathode in the same alkaline solution, as quantified by a delivered current density of 10 mA cm−2 at an overpotential of 400 mV for each of the oxygen and hydrogen evolution reactions. By driving two such identical electrodes with a solution-processed thin-film perovskite photovoltaic assembly, a wired artificial-leaf device is obtained that features a Faradaic H2 evolution efficiency of 100%, and a solar-to-hydrogen conversion efficiency of 6.2%. A detailed cost analysis is presented, which implies that the material-payback time of this device is of the order of 100 days.
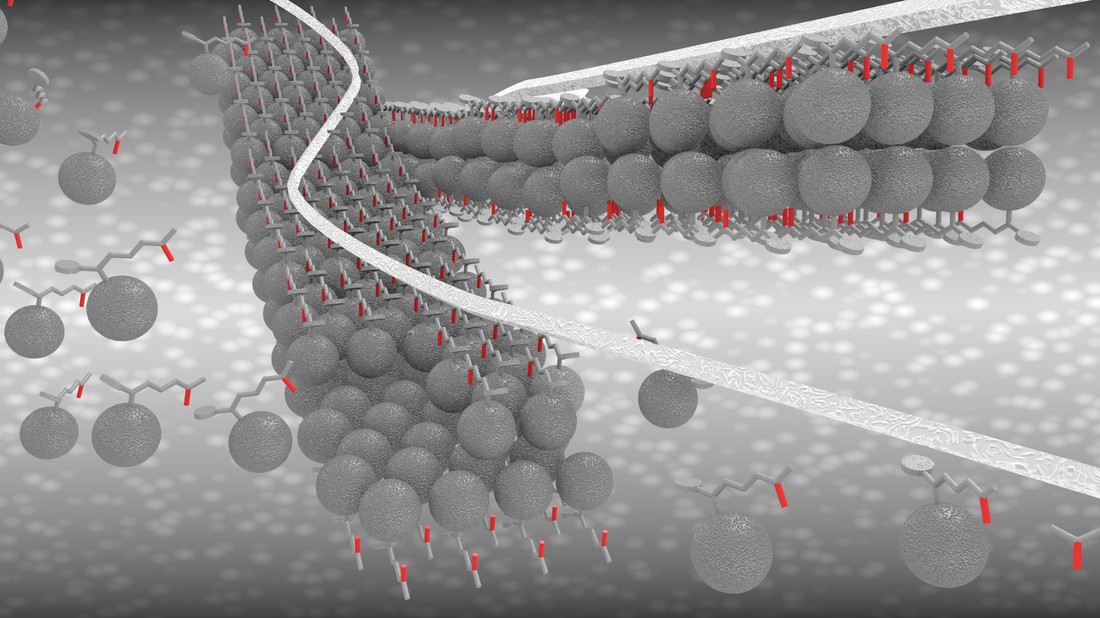
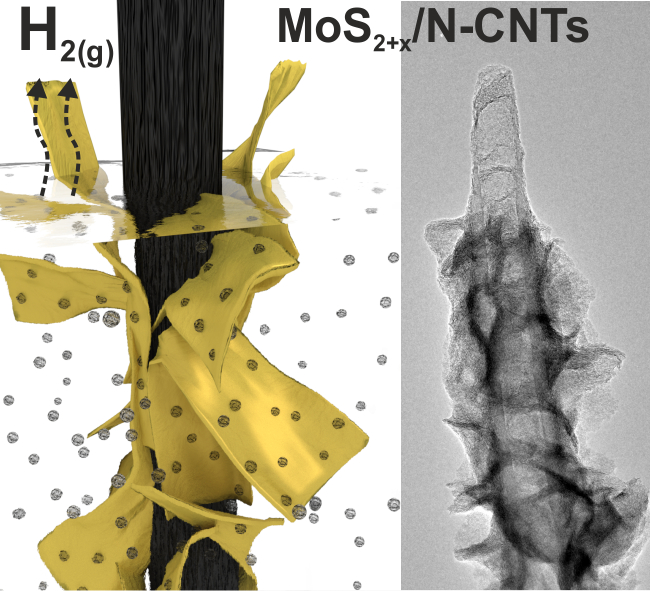
We report the production of a hybrid catalyst electrode comprising semicrystalline molybdenum sulfide (MoS2+x) attached on nitrogen-doped carbon nanotubes. The nitrogen-doping of the carbon nanotubes stabilizes a semicrystalline structure of MoS2+x with a high exposure of active sites for HER resulting in enhanced catalytic activity. The results are published in the journal of Advanced Functional Materials. Joakim Ekspong, Tiva Sharifi, Andrey Shchukarev, Alexey Klechikov, Thomas Wågberg, and Eduardo Gracia-Espino. Adv. Funct. Mater, 2016. DOI: 10.1002/adfm.201601994 Abstract Finding an abundant and cost-effective electrocatalyst for the hydrogen evolution reaction (HER) is crucial for a global production of hydrogen from water electrolysis. This work reports an exceptionally large surface area hybrid catalyst electrode comprising semicrystalline molybdenum sulfide (MoS2+x) catalyst attached on a substrate based on nitrogen-doped carbon nanotubes (N-CNTs), which are directly grown on carbon fiber paper (CP). It is shown here that nitrogen-doping of the carbon nanotubes improves the anchoring of MoS2+x catalyst compared to undoped carbon nanotubes and concurrently stabilizes a semicrystalline structure of MoS2+x with a high exposure of active sites for HER. The well-connected constituents of the hybrid catalyst are shown to facilitate electron transport and as a result of the good attributes, the MoS2+x/N-CNT/CP electrode exhibits an onset potential of −135 mV for HER in 0.5 m H2SO4, a Tafel slope of 36 mV dec−1, and high stability at a current density of −10 mA cm−2.
|
Nano for Energy group
Categories
All
Featured publications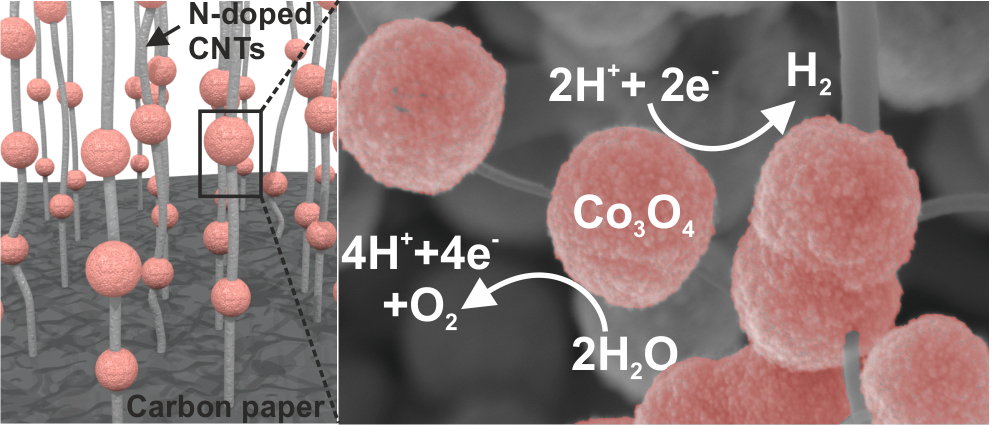
Comprehensive Study of an Earth-Abundant Bifunctional 3D Electrode for Efficient Water Electrolysis in Alkaline Medium.
ACS Appl. Mater. Interfaces, 2015, 7, 28148 
Fabrication of One-Dimensional Zigzag [6,6]-Phenyl-C61-Butyric Acid Methyl Ester Nanoribbons from Two-Dimensional Nanosheets.
ACS Nano, 2015, 9, 10516 
Hierarchical self-assembled structures based on nitrogen-doped carbon nanotubes as advanced negative electrodes for Li-ion batteries and 3D microbatteries.
J. P. Sources, 2015, 279, 581 .Self-Assembly Synthesis of Decorated Nitrogen-Doped Carbon Nanotubes with ZnO Nanoparticles: Anchoring Mechanism and the Effects of Sulfur.
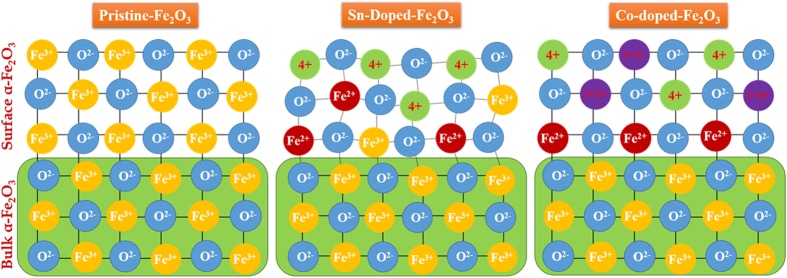
J. Phys. Chem. C, 120, 27849 (2016) Sn/Be Sequentially co-doped Hematite Photoanodes for Enhanced Photoelectrochemical Water Oxidation: Effect of Be2+ as co-dopant.
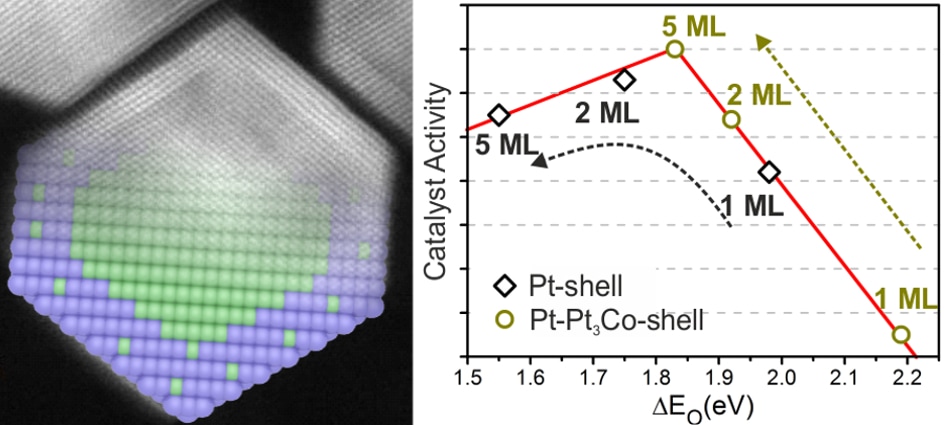
Sci Rep. 2016; 6: 23183. Atomistic understanding of the origin of high oxygen reduction electrocatalytic activity of cuboctahedral Pt3Co–Pt core–shell nanoparticles.
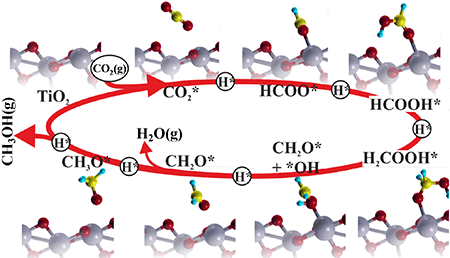
Catal. Sci. Technol., 2016, 6, 1393-1401 Photocatalytic reduction of CO2 with H2O over modified TiO2 nanofibers: Understanding the reduction pathway.
Nano Res. (2016) 9: 1956. |
 RSS Feed
RSS Feed