|
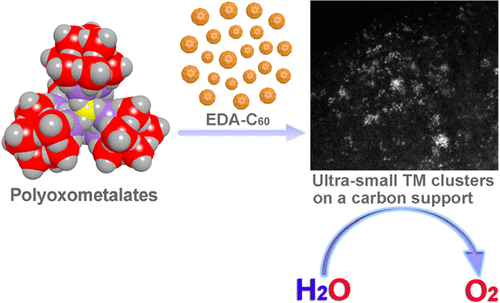
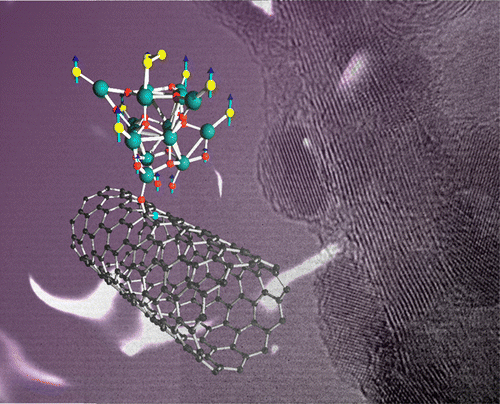
Here we report the use of a molecule-to-cluster strategy for preparing ultrasmall (~8 Å) trimetallic Co-Fe-W clusters with high efficiency for the OER in alkaline media. Besides the excellent performance as OER catalyst, our molecule-to-cluster strategy allows to achieve well-defined transition-metal clusters in the subnanometer regime. Xin-Bao Han, Xing-Yan Tang, Yue Lin, Eduardo Gracia-Espino, San-Gui Liu, Hai-Wei Liang, Guang-Zhi Hu, Xin-Jing Zhao, Hong-Gang Liao, Yuan-Zhi Tan, Thomas Wagberg, Su-Yuan Xie, Lan-Sun Zheng "Ultrasmall Abundant Metal-Based Clusters as Oxygen-Evolving Catalysts" J. Am. Chem. Soc. (2019) DOI: 10.1021/jacs.8b09076 Abstract The oxygen evolution reaction is a crucial step in water electrolysis to develop clean and renewable energy. Although noble metal-based catalysts have demonstrated high activity for the oxygen evolution reaction, their application is limited by their high cost and low availability. Here we report the use of a molecule-to-cluster strategy for preparing ultrasmall trimetallic clusters by using the polyoxometalate molecule as a precursor. Ultrafine (0.8 nm) transition-metal clusters with controllable chemical composition are obtained. The transition-metal clusters enable highly efficient oxygen evolution through water electrolysis in alkaline media, manifested by an overpotential of 192 mV at 10 mA cm–2, a low Tafel slope of 36 mV dec–1, and long-term stability for 30 h of electrolysis. We note, however, that besides the excellent performance as an oxygen evolution catalyst, our molecule-to-cluster strategy provides a means to achieve well-defined transition-metal clusters in the subnanometer regime, which potentially can have an impact on several other applications.
0 Comments
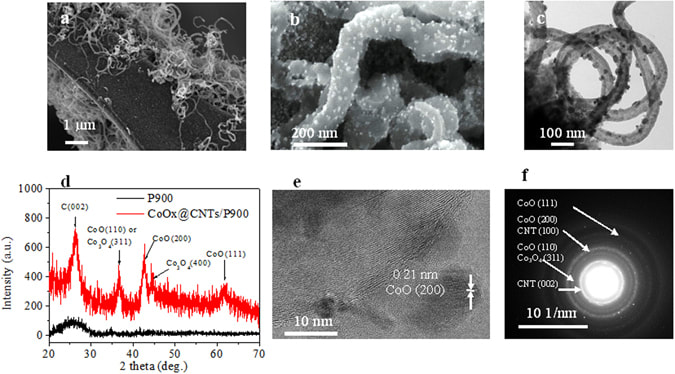
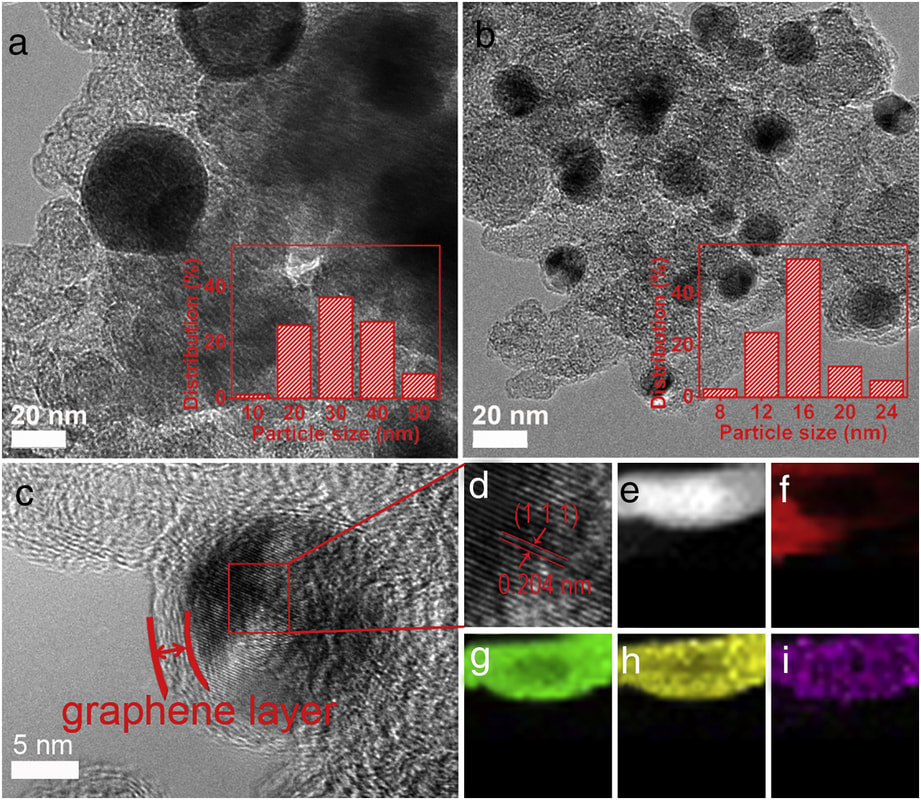
Hierarchical macroporous carbon foam decorated with cobalt oxide nanoparticles exhibit excellent performance for oxygen evolution reaction (OER). The observed electrocatalytic performance is rationalized by the overall 3D macroporous structure and with the firmly integrated CNTs directly grown on the foam. The work is a collaboration with Prof. Mikkola (Umeå, Sweden). The results are published in the journal of Scientific Reports. Tung Ngoc Pham, Tiva Sharifi, Robin Sandström, William Siljebo, Andrey Shchukarev, Krisztian Kordas, Thomas Wågberg, and Jyri-Pekka Mikkola Scientific Reports, 7, 6112 (2017) (Download) AbstractHerein we report a 3D heterostructure comprising a hierarchical macroporous carbon foam that incorporates mesoporous carbon nanotubes decorated with cobalt oxide nanoparticles as an unique and highly efficient electrode material for the oxygen evolution reaction (OER) in electrocatalytic water splitting. The best performing electrode material showed high stability after 10 h, at constant potential of 1.7 V vs. RHE (reversible hydrogen electrode) in a 0.1 M KOH solution and high electrocatalytic activity in OER with low overpotential (0.38 V vs RHE at 10 mA cm−2). The excellent electrocatalytic performance of the electrode is rationalized by the overall 3D macroporous structure and with the firmly integrated CNTs directly grown on the foam, resulting in a large specific surface area, good electrical conductivity, as well as an efficient electrolyte transport into the whole electrode matrix concurrent with an ability to quickly dispose oxygen bubbles into the electrolyte. The eminent properties of the three-dimensional structured carbon matrix, which can be synthesized through a simple, scalable and cost effective pyrolysis process show that it has potential to be implemented in large-scale water electrolysis systems.
This work was performed in collaboration with Prof. Guangzhi Hu from the University of Chinese Academy of Sciences. Here we report the microwave-assisted synthesis of Mo-doped FeNi3 nanoparticles as excellent oxygen evolution electrocatalyst. Our results were published in the journal of Electrochemistry Communications. Hangjia Shen, Eduardo Gracia-Espino, Le Wang, Dan feng Qin, Sanshuang Gao, Xamxikamar Mamat, Wei Ren, Thomas Wågberg, Guangzhi Hu. Electrochem. Commun. 81, 116-119 (2017) Abstract Oxygen evolution reaction (OER) plays a pivotal role in water-splitting. Here, we report a facile method to synthesize multimetal supported on commercial carbon black via a time-saving microwave process. Crystalline FeNi3 nanoparticles homogeneously doped with Mo are formed via a microwave treatment and activated to metal oxyhydroxide in-situ during cyclic voltammetry test with overpotential of only 280 mV at 10 mA cm− 2 for OER in alkaline electrolyte, outperforming RuO2. Our synthesis methodology is a promising alternative for large-scale production, delivering a valuable contribution to catalyst preparation and electrocatalytic water oxidation research.
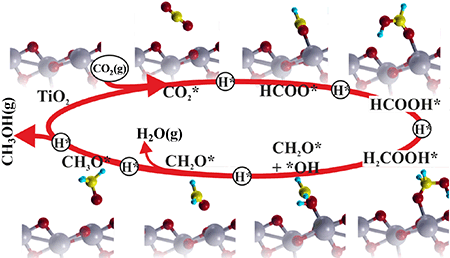
The photocatalytic performance of nanosized metal (Pt or Pd)-decorated TiO2 nanofibers (NFs) with CdSe quantum dots were tested for activation and reduction of CO2 under UV-B light. The CO2 photoreduction mechanism is proposed to take place via a hydrogenation pathway from first principles calculations. This work was performed in collaboration with the University of Oulu and the Åbo Akademi University, the manuscript is published in the journal of Nano Research. Anjana Sarkar, Eduardo Gracia-Espino, Thomas Wågberg, Andrey Shchukarev, Melinda Mohl, Anne-Riikka Rautio, Olli Pitkänen, Tiva Sharifi, Krisztian Kordas, Jyri-Pekka Mikkola. Nano Research, 2016 DOI:10.1007/s12274-016-1087-9 AbstractNanosized metal (Pt or Pd)-decorated TiO2 nanofibers (NFs) were synthesized by a wet impregnation method. CdSe quantum dots (QDs) were then anchored onto the metal-decorated TiO2 NFs. The photocatalytic performance of these catalysts was tested for activation and reduction of CO2 under UV-B light. Gas chromatographic analysis indicated the formation of methanol, formic acid, and methyl formate as the primary products. In the absence of CdSe QDs, Pd-decorated TiO2 NFs were found to exhibit enhanced performance compared to Pt-decorated TiO2 NFs for methanol production. However, in the presence of CdSe, Pt-decorated TiO2 NFs exhibited higher selectivity for methanol, typically producing ∼90 ppmg−1·h−1 methanol. The CO2 photoreduction mechanism is proposed to take place via a hydrogenation pathway from first principles calculations, which complement the experimental observations.
|
Nano for Energy group
Categories
All
Featured publications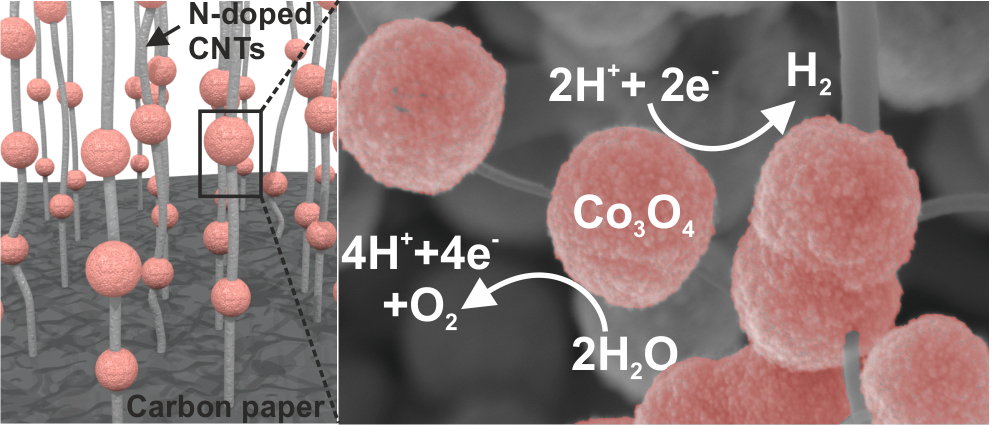
Comprehensive Study of an Earth-Abundant Bifunctional 3D Electrode for Efficient Water Electrolysis in Alkaline Medium.
ACS Appl. Mater. Interfaces, 2015, 7, 28148 
Fabrication of One-Dimensional Zigzag [6,6]-Phenyl-C61-Butyric Acid Methyl Ester Nanoribbons from Two-Dimensional Nanosheets.
ACS Nano, 2015, 9, 10516 
Hierarchical self-assembled structures based on nitrogen-doped carbon nanotubes as advanced negative electrodes for Li-ion batteries and 3D microbatteries.
J. P. Sources, 2015, 279, 581 .Self-Assembly Synthesis of Decorated Nitrogen-Doped Carbon Nanotubes with ZnO Nanoparticles: Anchoring Mechanism and the Effects of Sulfur.
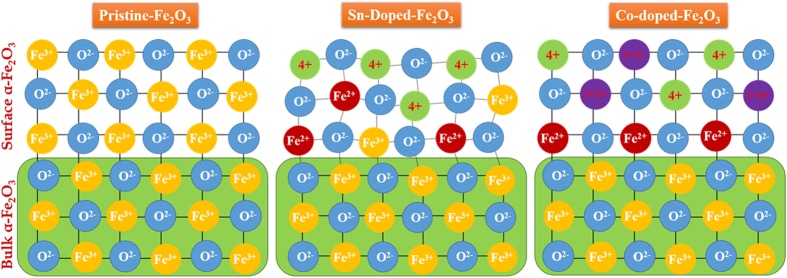
J. Phys. Chem. C, 120, 27849 (2016) Sn/Be Sequentially co-doped Hematite Photoanodes for Enhanced Photoelectrochemical Water Oxidation: Effect of Be2+ as co-dopant.
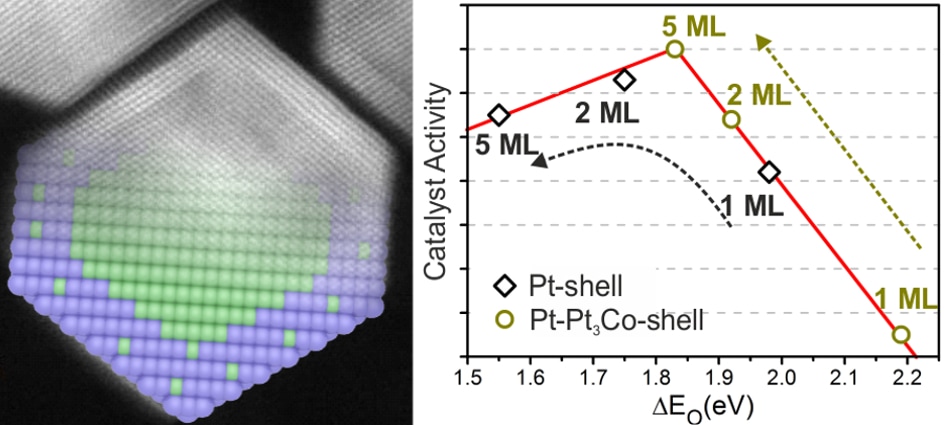
Sci Rep. 2016; 6: 23183. Atomistic understanding of the origin of high oxygen reduction electrocatalytic activity of cuboctahedral Pt3Co–Pt core–shell nanoparticles.
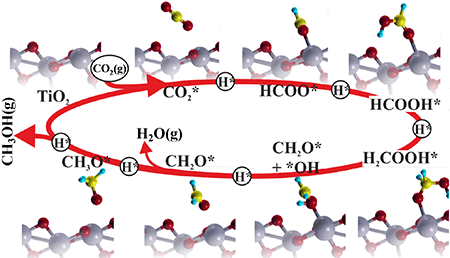
Catal. Sci. Technol., 2016, 6, 1393-1401 Photocatalytic reduction of CO2 with H2O over modified TiO2 nanofibers: Understanding the reduction pathway.
Nano Res. (2016) 9: 1956. |
 RSS Feed
RSS Feed