|
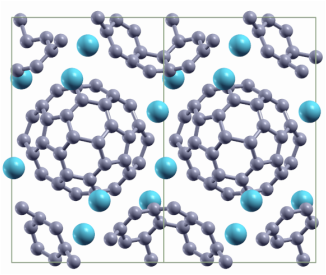
Here we performed calorimetric measurements of Na4C60 and Li4C60 crystals, the results are published in the Journal of Chemical Physics. Akira Inaba, Yuji Miyazaki, Paweł P. Michałowski, Eduardo Gracia-Espino, Bertil Sundqvist and Thomas Wågberg. J. Chem. Phys. 142, 164706 (2015) AbstractWe show specific heat data for Na4C60 and Li4C60 in the range 0.4-350 K for samples characterized by Raman spectroscopy and X-ray diffraction. At high temperatures, the two different polymer structures have very similar specific heats both in absolute values and in general trend. The specific heat data are compared with data for undoped polymeric and pristine C60. At high temperatures, a difference in specific heat between the intercalated and undoped C60 polymers of 100 J K−1 mol−1 is observed, in agreement with the Dulong-Petit law. At low temperatures, the specific heat data for Li4C60 and Na4C60 are modified by the stiffening of vibrational and librational molecular motion induced by the polymer bonds. The covalent twin bonds in Li4C60 affect these motions to a somewhat higher degree than the single intermolecular bonds in Na4C60. Below 1 K, the specific heats of both materials become linear in temperature, as expected from the effective dimensionality of the structure. The contribution to the total specific heat from the inserted metal ions can be well described by Einstein functions with T E = 386 K for Li4C60 and T E = 120 K for Na4C60, but for both materials we also observe a Schottky-type contribution corresponding to a first approximation to a two-level system with ΔE = 9.3 meV for Li4C60 and 3.1 meV for Na4C60, probably associated with jumps between closely spaced energy levels inside “octahedral-type” ionic sites. Static magnetic fields up to 9 T had very small effects on the specific heat below 10 K.
0 Comments
Your comment will be posted after it is approved.
Leave a Reply. |
Nano for Energy group
Categories
All
Featured publications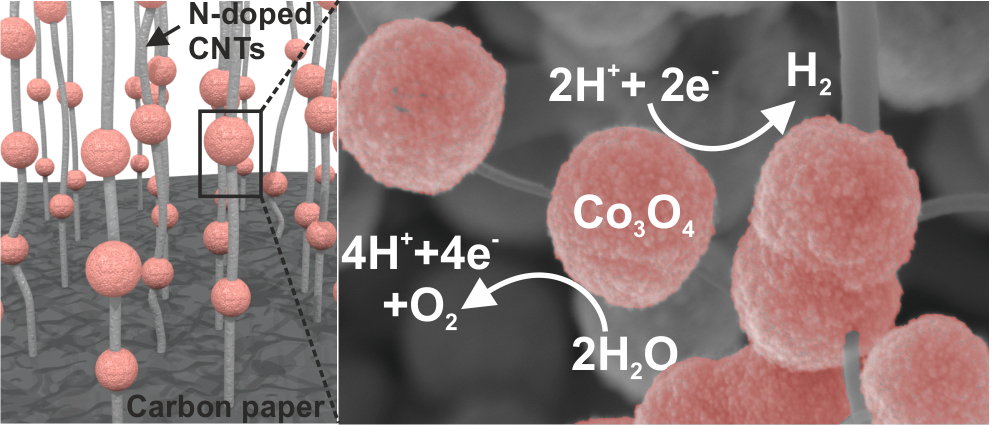
Comprehensive Study of an Earth-Abundant Bifunctional 3D Electrode for Efficient Water Electrolysis in Alkaline Medium.
ACS Appl. Mater. Interfaces, 2015, 7, 28148 
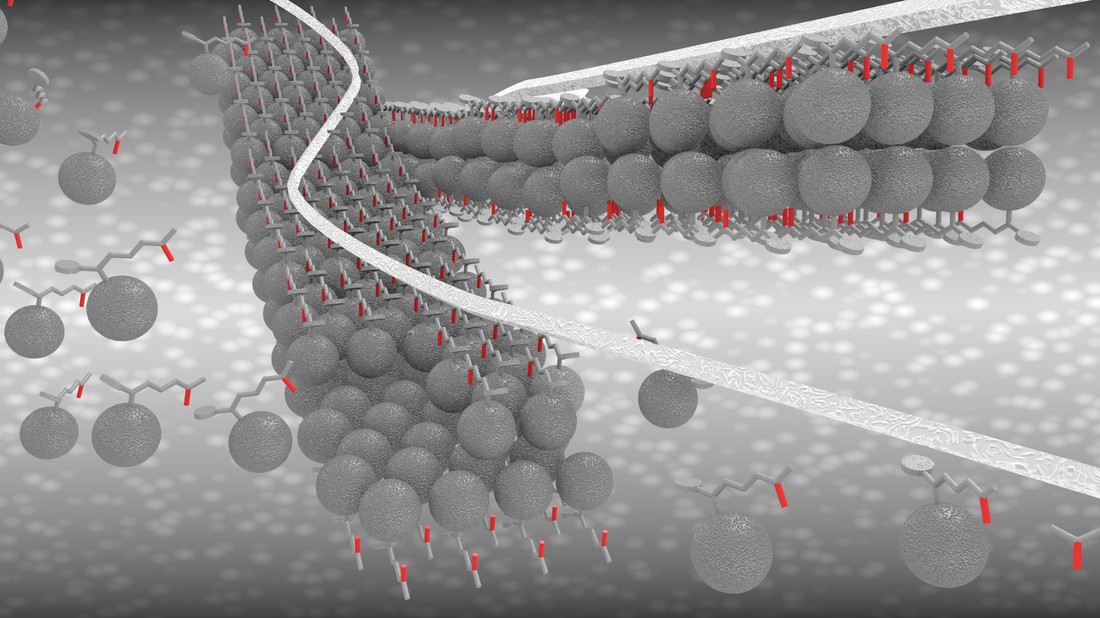
Fabrication of One-Dimensional Zigzag [6,6]-Phenyl-C61-Butyric Acid Methyl Ester Nanoribbons from Two-Dimensional Nanosheets.
ACS Nano, 2015, 9, 10516 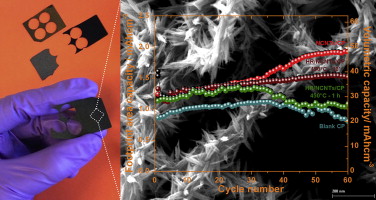
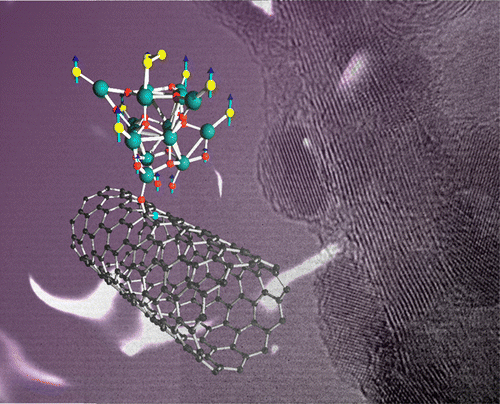
Hierarchical self-assembled structures based on nitrogen-doped carbon nanotubes as advanced negative electrodes for Li-ion batteries and 3D microbatteries.
J. P. Sources, 2015, 279, 581 .Self-Assembly Synthesis of Decorated Nitrogen-Doped Carbon Nanotubes with ZnO Nanoparticles: Anchoring Mechanism and the Effects of Sulfur.
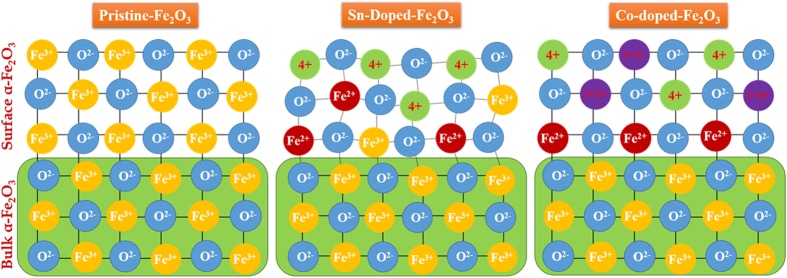
J. Phys. Chem. C, 120, 27849 (2016) Sn/Be Sequentially co-doped Hematite Photoanodes for Enhanced Photoelectrochemical Water Oxidation: Effect of Be2+ as co-dopant.
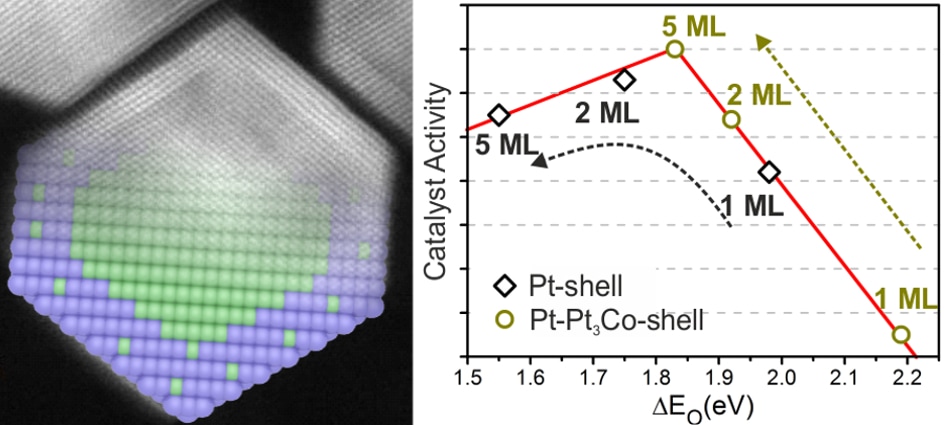
Sci Rep. 2016; 6: 23183. Atomistic understanding of the origin of high oxygen reduction electrocatalytic activity of cuboctahedral Pt3Co–Pt core–shell nanoparticles.
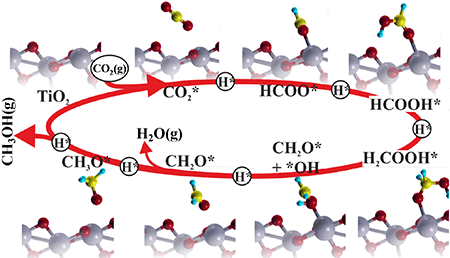
Catal. Sci. Technol., 2016, 6, 1393-1401 Photocatalytic reduction of CO2 with H2O over modified TiO2 nanofibers: Understanding the reduction pathway.
Nano Res. (2016) 9: 1956. |
 RSS Feed
RSS Feed