|
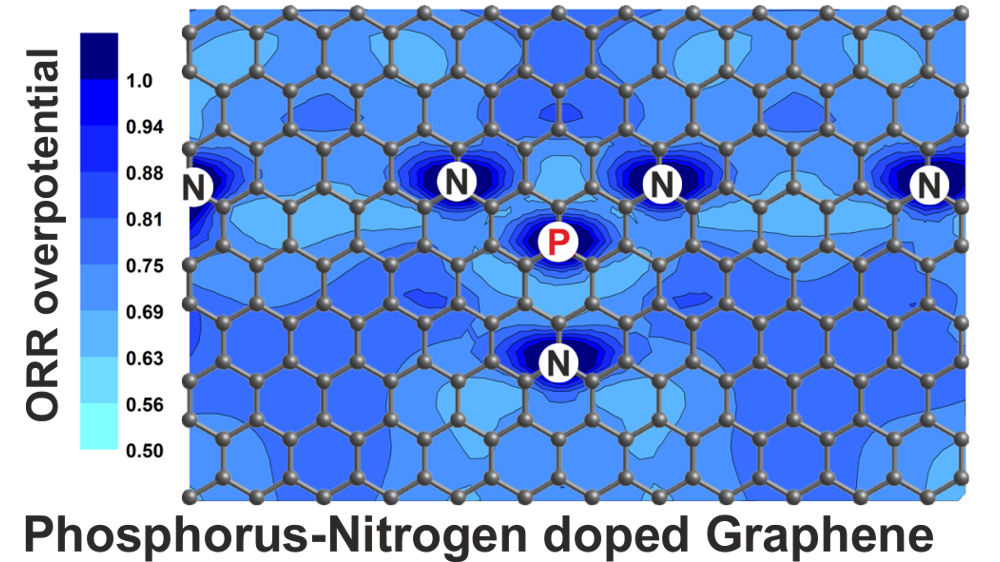
In our most recent publication we performed ab initio calculations to construct ORR overpotential maps and describe the availability and spatial distribution of catalytic active sites on phosphorus-nitrogen co-doped graphene. The results are published in the Journal of Physical Chemistry C. Eduardo Gracia-Espino. J. Phys. Chem. C, 120, 27849–27857 (2016) DOI: 10.1021/acs.jpcc.6b09425 Abstract Ab initio calculations are performed to investigate how the simultaneous introduction of phosphorus and nitrogen into graphene modifies the availability and spatial distribution of catalytic active sites for oxygen reduction reaction (ORR). A phosphoryl group (R3-P=O) is selected as a representative for the phosphorus doping, and the ORR is studied under alkaline conditions where a 4e- mechanism is used to determine the limiting step and overpotential (ηORR) along the entire graphene surface. A scanning procedure is used to construct ηORR maps for pristine-, N-, P-, and diverse PN co-doped graphenes. The results indicate that a single N (P) atom activates up to 17 (3) C atoms, while the simultaneous introduction of P and N activates up to 55 C atoms equivalent to 57% of the surface. Additionally, PN co-doped graphenes reveals that the relative location of both dopants has significant effects on the ORR performance, where a P-N separation distance of at least 4 Å minimize the localization of electronic states on the neighboring C atoms and improves the quantity and distribution of active sites. The results shows the importance of designing synthesis procedures to control the dopant concentration and spatial distribution to maximize the number of active sites. Furthermore, the ηORR maps reveal features that could be obtained by scanning tunneling microscopy allowing to experimentally identify and possibly quantify the catalytic active sites on carbon-based materials.
0 Comments
Recently our paper "Toward a Low-Cost Artificial Leaf: Driving Carbon-Based and Bifunctional Catalyst Electrodes with Solution-Processed Perovskite Photovoltaics" published in Advanced Energy Materials was highlighted on the Inside Front Cover, and here it is: Thomas Wågberg, Ludvig Edman, and co-workers present in article number 1600738 an artificial-leaf device comprising lightweight electrocatalyst electrodes powered by solution-processed perovskite photovoltaics. The electrocatalyst, comprising NiCo2O4 nanorods anchored onto carbon paper via nitrogen-doped carbon nanotubes, operates efficiently as both anode and cathode in alkaline solution. The wired artificial leaf can be very low cost and features a solar-to-hydrogen efficiency of 6.2%.
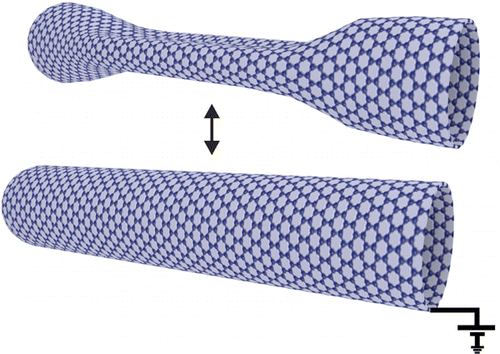
Source: Advanced Energy Material, inside cover, volume 6, issue 20, October 26, 2016. Umeå University has published a press release of our latest article about "Electrostatically Driven Nanoballoon Actuator" reported in the journal of Nano Letters. Here is a glance of the press release: Molecular sized machines could in the future be used to control important mechanisms in the body. In a recent study, researchers at University of California, Berkeley and Umeå University show how a nanoballoon comprising a single carbon molecule ten thousand times thinner than a human hair can be controlled electrostatically to switch between an inflated and a collapsed state. Schematic drawing of two carbon nanotubes, one in inflated state (cylindrical tube to the right) and one in collapsed state (flattened tube to the left). The transition between the two states can be controlled by applying a small voltage which charges the tube electrostatically and thereby changes the state from a collapsed state to an inflated. The applied voltage is visualized by two tip contacts touching each tube. In the image the tubes are connected to two pistons to envision that the phase change of the tubes could set an imaginary nanomachine in motion.
Hamid Reza Barzegar, Aiming Yan, Sinisa Coh, Eduardo Gracia-Espino, Gabriel Dunn, Thomas Wågberg, Steven G. Louie, Marvin L. Cohen, and Alex Zettl. Nano Lett., DOI: 10.1021/acs.nanolett.6b02394 This time we show that collapsed carbon nanotubes can be easily and reliable cycled between their inflated and collapsed states by electrically charging the nanotubes forming nanoballoon actuators. The work has been developed in close collaboration with the research group of Prof. Zettl from the University of California, Berkeley. The results have been published in the journal of Nano Letters. Hamid Reza Barzegar, Aiming Yan, Sinisa Coh, Eduardo Gracia-Espino, Gabriel Dunn, Thomas Wågberg, Steven G. Louie, Marvin L. Cohen, and Alex Zettl. Nano Lett., DOI: 10.1021/acs.nanolett.6b02394 Abstract We demonstrate an inflatable nanoballoon actuator based on geometrical transitions between the inflated (cylindrical) and collapsed (flattened) forms of a carbon nanotube. In situ transmission electron microscopy experiments employing a nanoelectromechanical manipulator show that a collapsed carbon nanotube can be reinflated by electrically charging the nanotube, thus realizing an electrostatically driven nanoballoon actuator. We find that the tube actuator can be reliably cycled with only modest control voltages (few volts) with no apparent wear or fatigue. A complementary theoretical analysis identifies critical parameters for nanotube nanoballoon actuation.
|
Nano for Energy group
Categories
All
Featured publications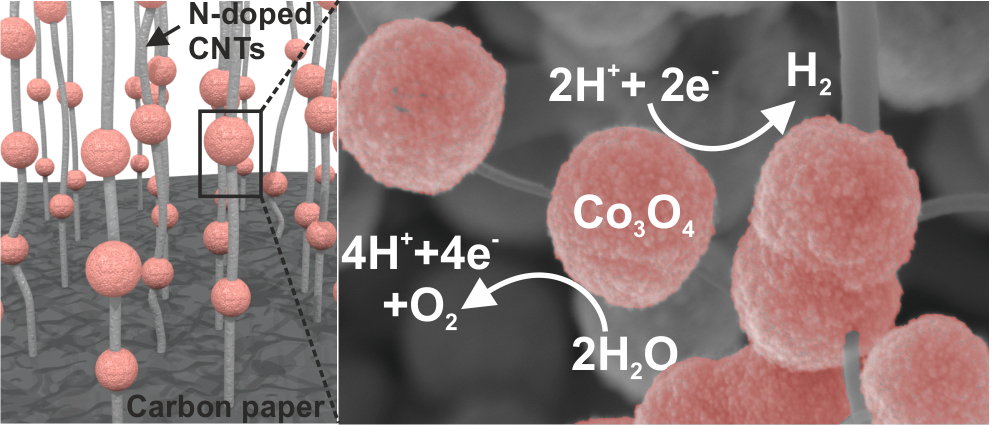
Comprehensive Study of an Earth-Abundant Bifunctional 3D Electrode for Efficient Water Electrolysis in Alkaline Medium.
ACS Appl. Mater. Interfaces, 2015, 7, 28148 
Fabrication of One-Dimensional Zigzag [6,6]-Phenyl-C61-Butyric Acid Methyl Ester Nanoribbons from Two-Dimensional Nanosheets.
ACS Nano, 2015, 9, 10516 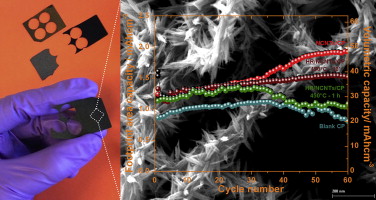
Hierarchical self-assembled structures based on nitrogen-doped carbon nanotubes as advanced negative electrodes for Li-ion batteries and 3D microbatteries.
J. P. Sources, 2015, 279, 581 .Self-Assembly Synthesis of Decorated Nitrogen-Doped Carbon Nanotubes with ZnO Nanoparticles: Anchoring Mechanism and the Effects of Sulfur.
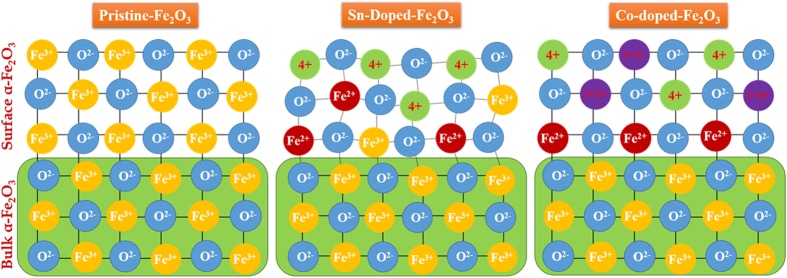
J. Phys. Chem. C, 120, 27849 (2016) Sn/Be Sequentially co-doped Hematite Photoanodes for Enhanced Photoelectrochemical Water Oxidation: Effect of Be2+ as co-dopant.
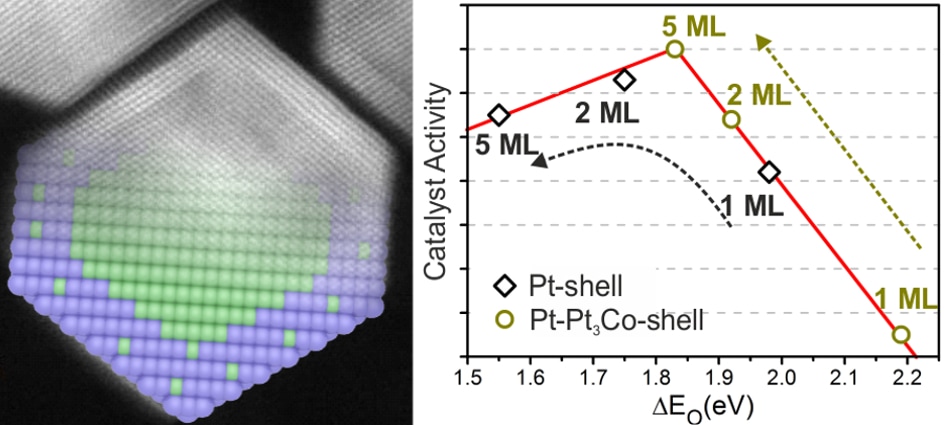
Sci Rep. 2016; 6: 23183. Atomistic understanding of the origin of high oxygen reduction electrocatalytic activity of cuboctahedral Pt3Co–Pt core–shell nanoparticles.
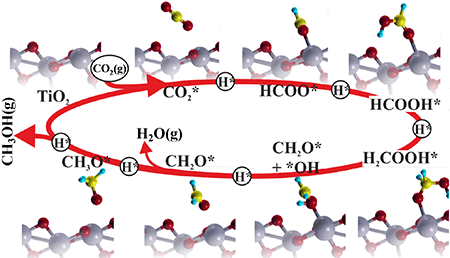
Catal. Sci. Technol., 2016, 6, 1393-1401 Photocatalytic reduction of CO2 with H2O over modified TiO2 nanofibers: Understanding the reduction pathway.
Nano Res. (2016) 9: 1956. |


 RSS Feed
RSS Feed